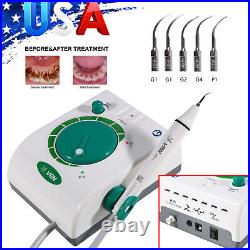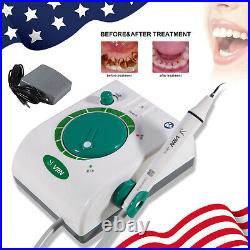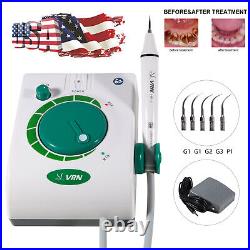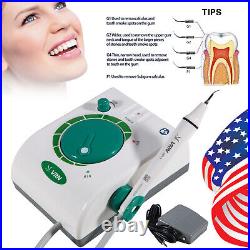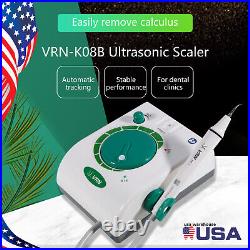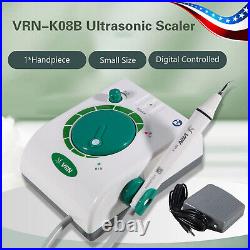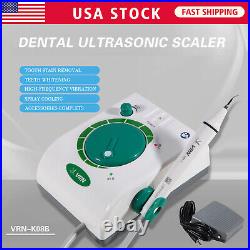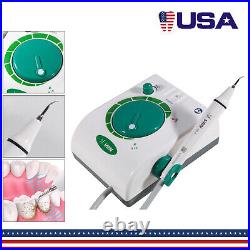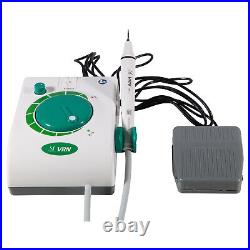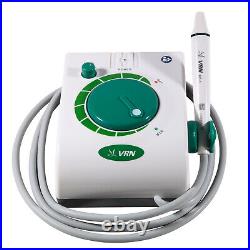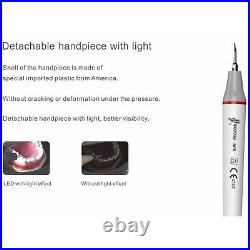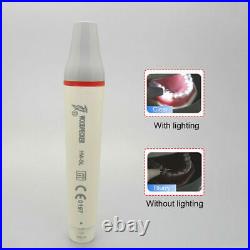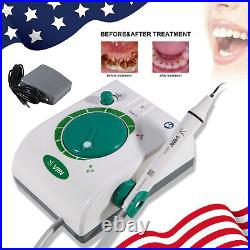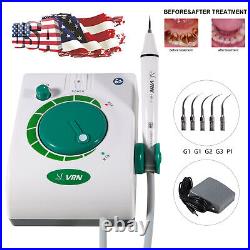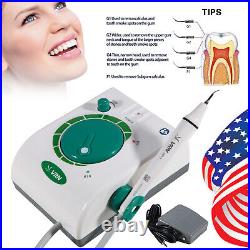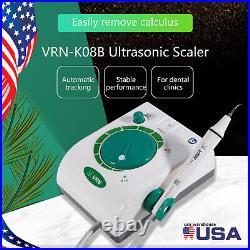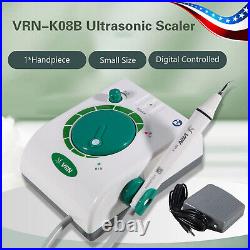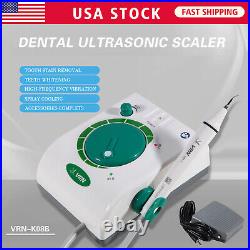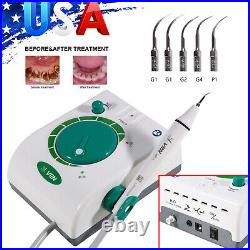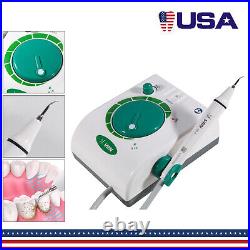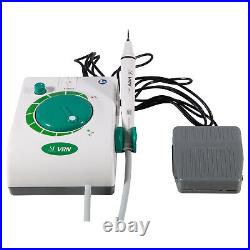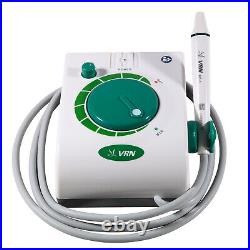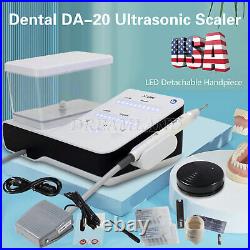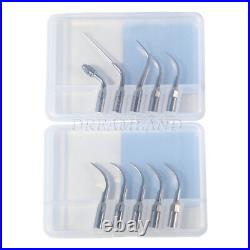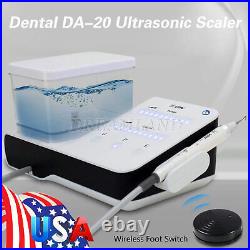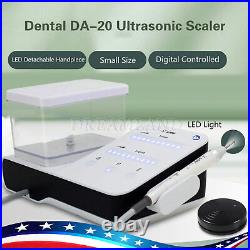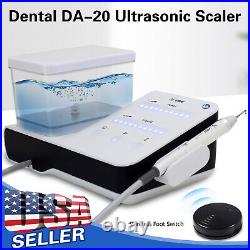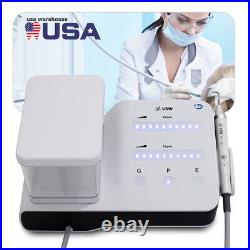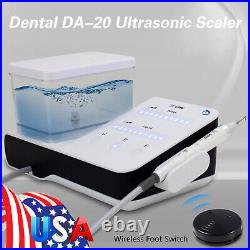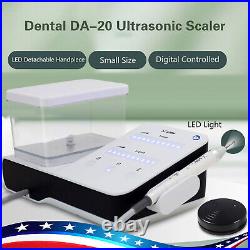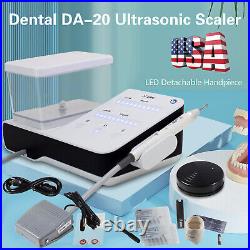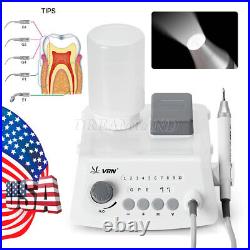



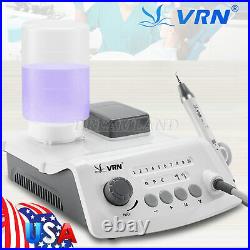




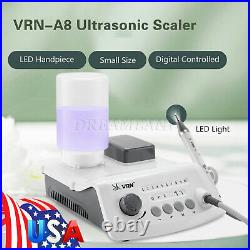



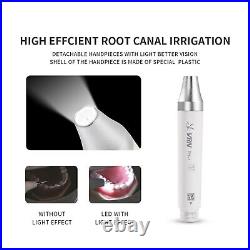





Dental Wireless Control Ultrasonic Scaler with LED Detachable Handpiece. As a new strategic product of VRN for the high-end ultrasonic scaler market, VRN-A8 absorbed the excellent quality as VRN ever did before, adopting the exclusive patent technology – wireless control, with multiple functions, automatic water supply, intelligent vibration technology. It brings you a convenient, faster and more comfortable experience for treatment. Patented Liquid Pumps, Digital Controlled. Function: Scaling, Perio, Endo. Control: Wireless foot switch. Two Water Supply: Auto-water supply. Handpiece: LED detachable handpiece. Power supply:AC adapter: 100-240vac. For main unit: 30Vdc. Wireless control fool pedal battery: 1.5VX2. Primary vibration excursion of the tip. (a)Minimum, 1um, Deviation -50%. Vibration frequency of the tip: 25-31KHz. Input power: 30VA- 48VA. Ultrasonic output power: 3W-20W. Period of use: 10 years. Fuse: 125V 1.5A. Water pressure: O1MPa-0.05MPa. Weight of main unit: 0.75kg. Weight of adapter: 0.25kg. Size: 230mmx 155mm x 56mm. 5 Tips: G1, G2, G4, P1, E1. The sale of this item may be subject to regulation by the U. Food and Drug Administration and state and local regulatory agencies. FDA for Curing Light: 510(K) Number: K192233, Regulation Number: 21 CFR 872.6070, Product Code: EBZ. FDA for Handpiece: 510(K) Number: K181691, Regulation Number: 872.4200, Classification Product Code: EFB, Subsequent Product Code: EGS. FDA for Scaler: 510(K) Number: K163414, Regulation Number: 872.4850, Product Code: ELC.